Original art for sale
Shop original and limited edition art, directly from artists around the world.
Clear all 
Rashna Hackett
Acrylic painting
94 x 136cm
£750
Victoria Obolensky
Acrylic painting
40 x 40cm
£495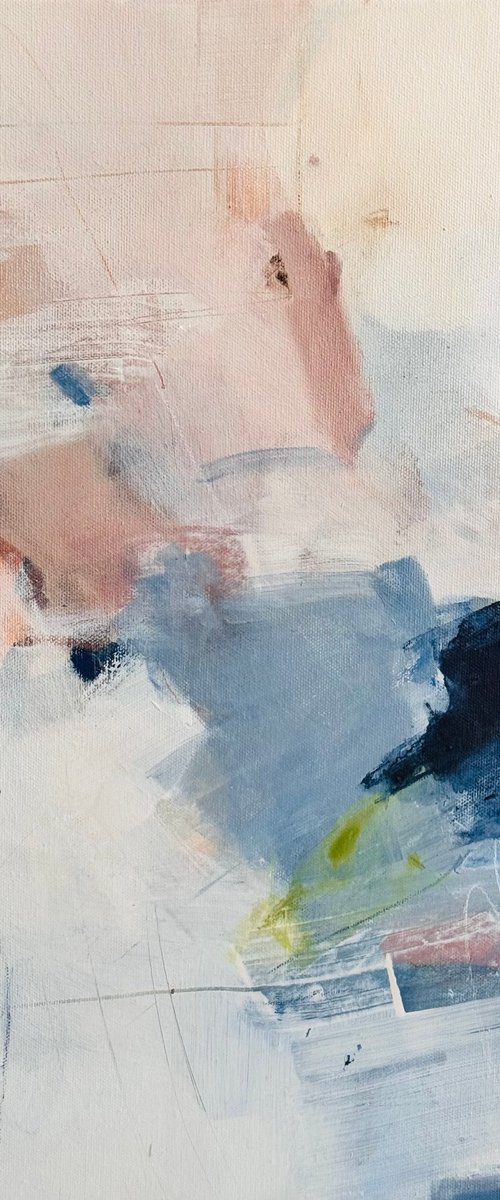
Arohika Verma
Acrylic painting
41 x 51cm
£625
Daria Gerasimova
Oil painting
70 x 60cm
£683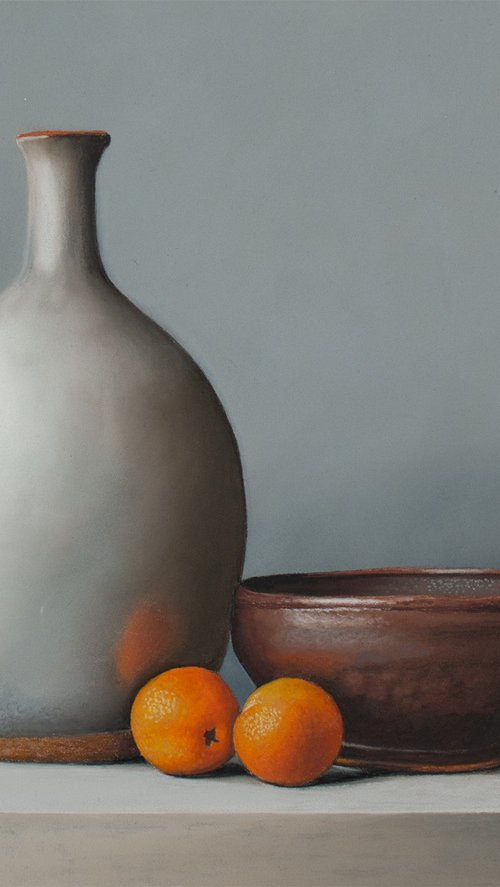
Dietrich Moravec
Pastel drawing
40 x 30cm
£427
Oleg Kaznacheiev
Watercolour
31 x 46cm
£597
Carlos Martín
Acrylic painting
25 x 25cm
£588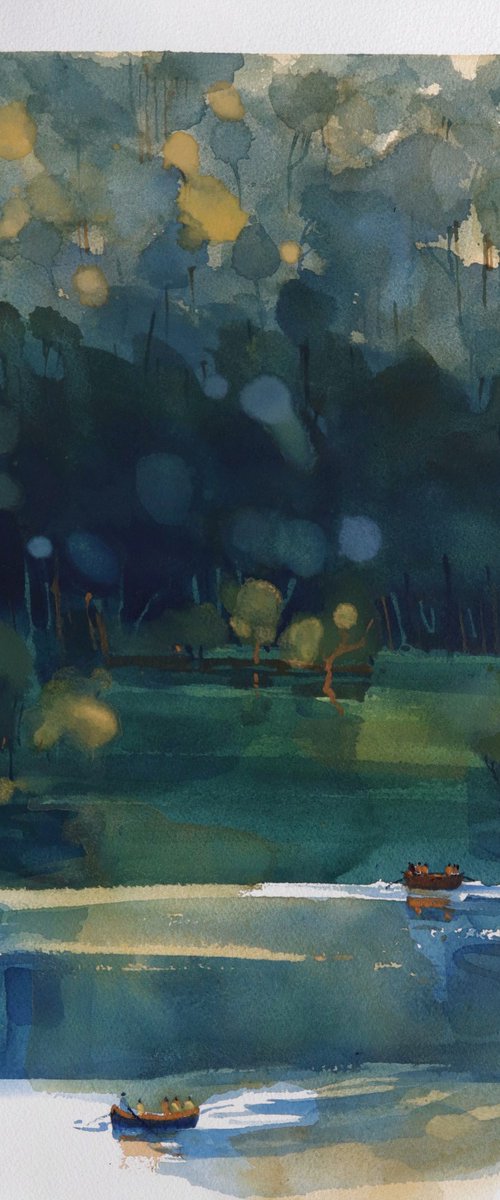
Prashant Prabhu
Watercolour
46 x 57cm
£574
Paul Cheng
Acrylic painting
61 x 76cm
£625
Igor Dubovoy
Oil painting
86 x 109cm
£3667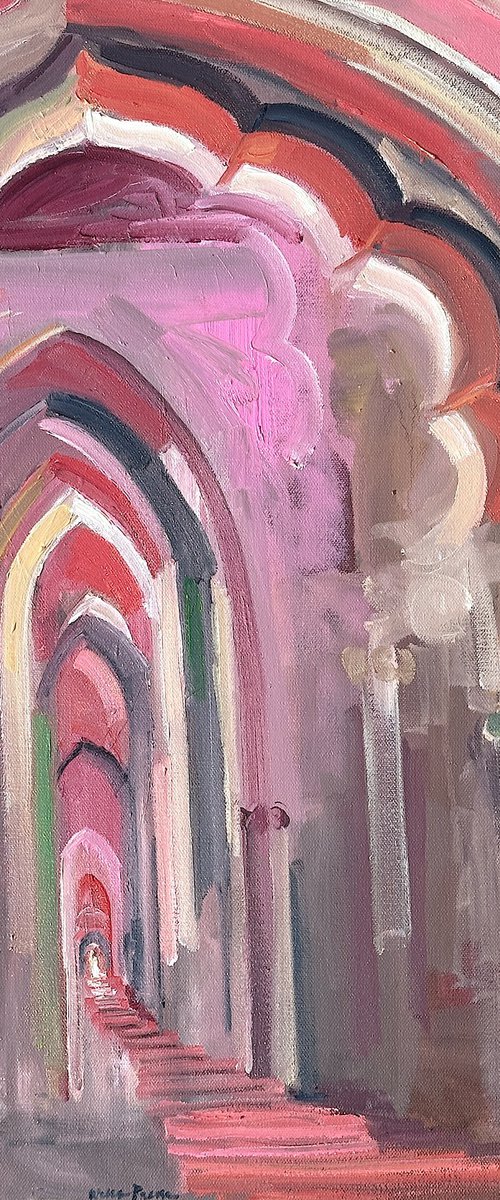
Arun Prem
Oil painting
51 x 61cm
£647
Teresa Tanner
Mixed-media painting
61 x 61cm
£700